|
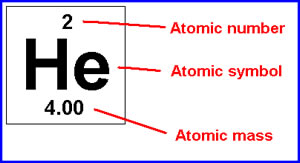
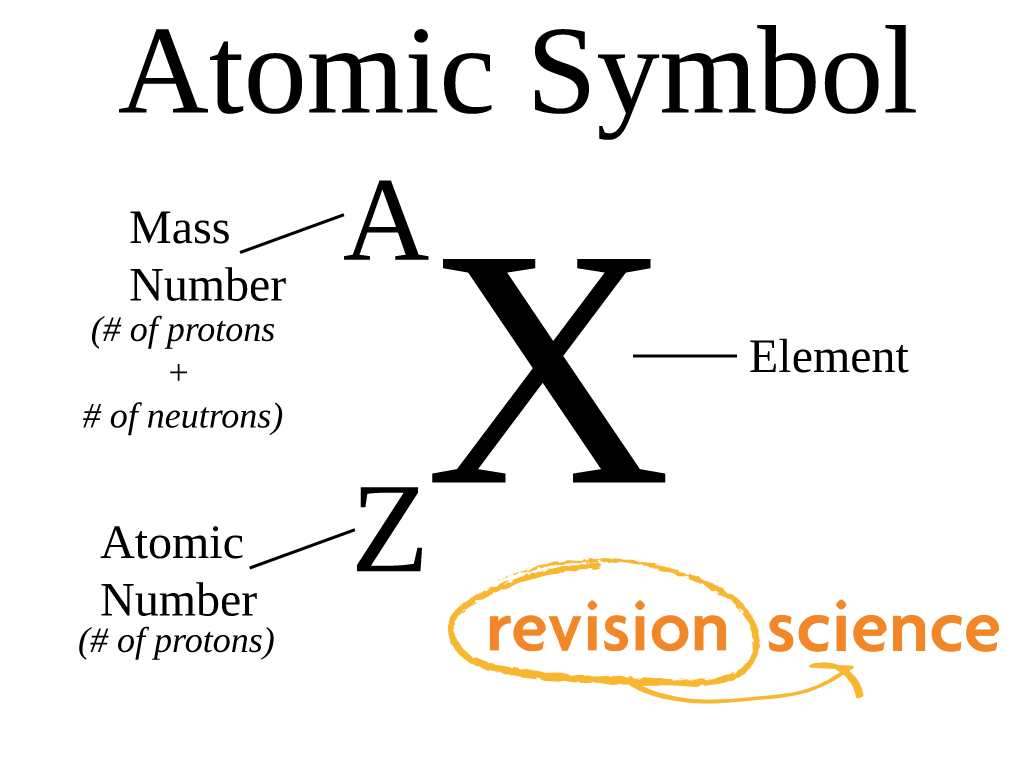
99.98% of all hydrogen is 1H 0.018% of all hydrogen is 2H 0. Each isotope has a different mass number.ġH has 1 proton. Hydrogen has three natural isotopes: 1H, 2H, and 3H. The mass number is a count of the total number of protons and neutrons in an atom's nucleus. Atomic mass is the weighted average mass of an atom of an element based on the relative natural abundanceof that element's isotopes. One is the average weight of an element and the other is the total number of nucleons in the atom's nucleus.Ītomic mass is also known as atomic weight. There is a difference between the meanings of the chemistry terms atomic mass and mass number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
